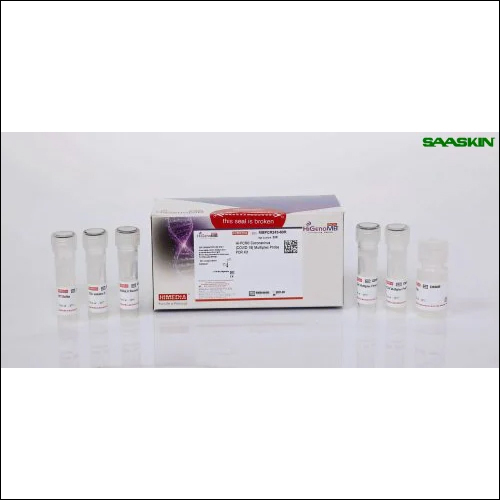
Hi-PCR Coronavirus (COVID-19) Multiplex Probe PCR Kit
Product Details:
- Instruments Type Molecular Diagnostic Kit
- Shelf Life 12 months from date of manufacture
- Accuracy High - Detects as low as 10 copies/reaction (depends on protocol)
- Function Detection of Coronavirus (COVID-19) RNA
- Features High sensitivity and specificity, Fast detection, Multiplexing for multiple gene targets, Ready-to-use master mix, Includes all necessary components
- Usage Type Professional Laboratory Use
- Measurement Range Qualitative detection
- Click to View more
X
Hi-PCR Coronavirus (COVID-19) Multiplex Probe PCR Kit Price And Quantity
- 1000 Box
- 25 µL per reaction
- Three viral genes: RdRp, N gene, and ORF1ab (as per kit description)
- As low as 10 copies per reaction
- Qualitative detection of novel coronavirus RNA in clinical laboratory settings
- Multiplex Probe Master Mix, Primer/Probe Mix for SARS-CoV-2, Positive and Negative Controls, Internal Control, Nuclease-free water
- Nasopharyngeal and oropharyngeal swabs; viral RNA from human samples
- Results in approximately 90 minutes
- Included for process validation
- Works with major Real-Time PCR platforms (e.g., ABI, BioRad, Roche)
- MBPCR262
- IVD CE Marked (as per manufacturer), for Research and Diagnostic Use
- Boxed kit with instruction manual
- 50 reactions (50 tests)
Hi-PCR Coronavirus (COVID-19) Multiplex Probe PCR Kit Product Specifications
- PCR Grade Reagents and Enzymes
- Qualitative detection
- Real-Time PCR (RT-PCR) with Multiplex Probe Detection
- Yes
- Store at -20C
- Kit Packaging Dimensions: 15 x 10 x 8 cm (approx.)
- Detection of Coronavirus (COVID-19) RNA
- Manual with Real-Time PCR Instrument
- Yes
- Molecular Diagnostic Kit
- N/A (Requires Laboratory PCR Thermocycler)
- No
- New
- Detection of SARS-CoV-2 (COVID-19) RNA in clinical samples
- High sensitivity and specificity, Fast detection, Multiplexing for multiple gene targets, Ready-to-use master mix, Includes all necessary components
- 12 months from date of manufacture
- High - Detects as low as 10 copies/reaction (depends on protocol)
- COVID-19 Multiplex Probe PCR Kit
- Kit with colored labels, colorless reagents
- Approx. 250 g per kit
- Professional Laboratory Use
- 25 µL per reaction
- Three viral genes: RdRp, N gene, and ORF1ab (as per kit description)
- As low as 10 copies per reaction
- Qualitative detection of novel coronavirus RNA in clinical laboratory settings
- Multiplex Probe Master Mix, Primer/Probe Mix for SARS-CoV-2, Positive and Negative Controls, Internal Control, Nuclease-free water
- Nasopharyngeal and oropharyngeal swabs; viral RNA from human samples
- Results in approximately 90 minutes
- Included for process validation
- Works with major Real-Time PCR platforms (e.g., ABI, BioRad, Roche)
- MBPCR262
- IVD CE Marked (as per manufacturer), for Research and Diagnostic Use
- Boxed kit with instruction manual
- 50 reactions (50 tests)
Product Description
Introducing the Featured Hi-PCR Coronavirus (COVID-19) Multiplex Probe PCR Kit, an Outstanding diagnostic solution at a Reduced Price for professional laboratories. This Exclusive kit delivers virtuoso accuracy, detecting as low as 10 copies per reaction using three viral targets: RdRp, N gene, and ORF1ab. The kit is IVD CE Marked, ensuring regulatory confidence. Equipped with all essential components-including multiplex probe master mix and controls-it is compatible with leading real-time PCR platforms. Results are achieved rapidly in approximately 90 minutes, supporting exceptional laboratory efficiency and reliability.
Exceptional Features & Competitive Advantages
The Hi-PCR Coronavirus (COVID-19) Multiplex Probe PCR Kit boasts superlative sensitivity and specificity, detecting SARS-CoV-2 RNA in both nasopharyngeal and oropharyngeal swab samples. Designed for professional laboratory use, it includes all necessary reagents, process validation controls, and delivers qualitative results fast. Its multiplex capability enables simultaneous identification of three viral genes, significantly improving throughput. Ready-to-use and portable, this kit ensures convenience and efficiency, distinguishing itself through accuracy and reduced turnaround time for diagnostic settings.
Certifications, Payment, and Domestic Availability
The Hi-PCR Multiplex PCR Kit is IVD CE Marked, confirming its rigorous compliance for clinical diagnostics. Payment terms are flexible, with prompt quotation on request to estimate your specific needs. Stock is ready for dispatch, ensuring minimal lead times for dealer, distributor, exporter, manufacturer, supplier, and wholesaler channels. Serving primarily the Indian domestic market, this kit's efficient supply chain guarantees trustworthy availability and consistent performance for healthcare and research institutions across the country.
Exceptional Features & Competitive Advantages
The Hi-PCR Coronavirus (COVID-19) Multiplex Probe PCR Kit boasts superlative sensitivity and specificity, detecting SARS-CoV-2 RNA in both nasopharyngeal and oropharyngeal swab samples. Designed for professional laboratory use, it includes all necessary reagents, process validation controls, and delivers qualitative results fast. Its multiplex capability enables simultaneous identification of three viral genes, significantly improving throughput. Ready-to-use and portable, this kit ensures convenience and efficiency, distinguishing itself through accuracy and reduced turnaround time for diagnostic settings.
Certifications, Payment, and Domestic Availability
The Hi-PCR Multiplex PCR Kit is IVD CE Marked, confirming its rigorous compliance for clinical diagnostics. Payment terms are flexible, with prompt quotation on request to estimate your specific needs. Stock is ready for dispatch, ensuring minimal lead times for dealer, distributor, exporter, manufacturer, supplier, and wholesaler channels. Serving primarily the Indian domestic market, this kit's efficient supply chain guarantees trustworthy availability and consistent performance for healthcare and research institutions across the country.
FAQ's of Hi-PCR Coronavirus (COVID-19) Multiplex Probe PCR Kit:
Q: How does the Hi-PCR Multiplex Probe PCR Kit optimize COVID-19 detection?
A: By targeting three separate viral genes-RdRp, N gene, and ORF1ab-the kit ensures precise identification and reduces the risk of false negatives, offering high analytical sensitivity at just 10 copies per reaction.Q: What components are included in the Hi-PCR Coronavirus PCR Kit?
A: The kit provides a multiplex probe master mix, primer/probe mixtures for SARS-CoV-2, positive and negative assay controls, nuclease-free water, an internal control for process validation, and a detailed instruction manual.Q: When can I expect to receive test results after running the assay?
A: Results can be obtained in approximately 90 minutes after preparing and running the test on a compatible real-time PCR instrument.Q: Where can this PCR kit be used and who is it intended for?
A: This kit is designed for use in professional clinical laboratory environments such as hospitals, diagnostic centers, and research labs in accordance with regulatory norms.Q: What is the process for handling and storing the kit?
A: Store the kit at -20C as specified, and ensure reagents remain in optimal condition before usage. All necessary instructions are included for smooth integration into laboratory workflow.Q: What are the main benefits of using this kit for COVID-19 diagnostics?
A: Key benefits include high sensitivity and specificity, reduced turnaround times, ready-to-use reagents, qualitative multiplex detection, and compliance with IVD CE Mark regulations, ensuring reliable and efficient results.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Medical IVD' category
 |
SAASKIN CORPORATION PRIVATE LIMITED
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |