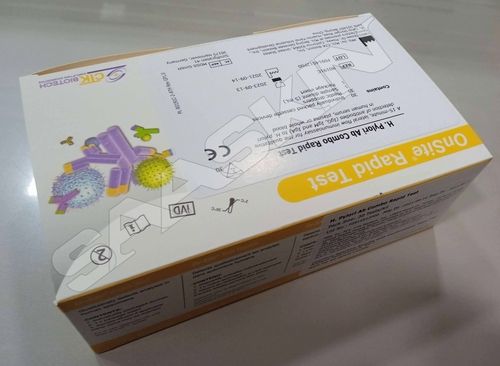
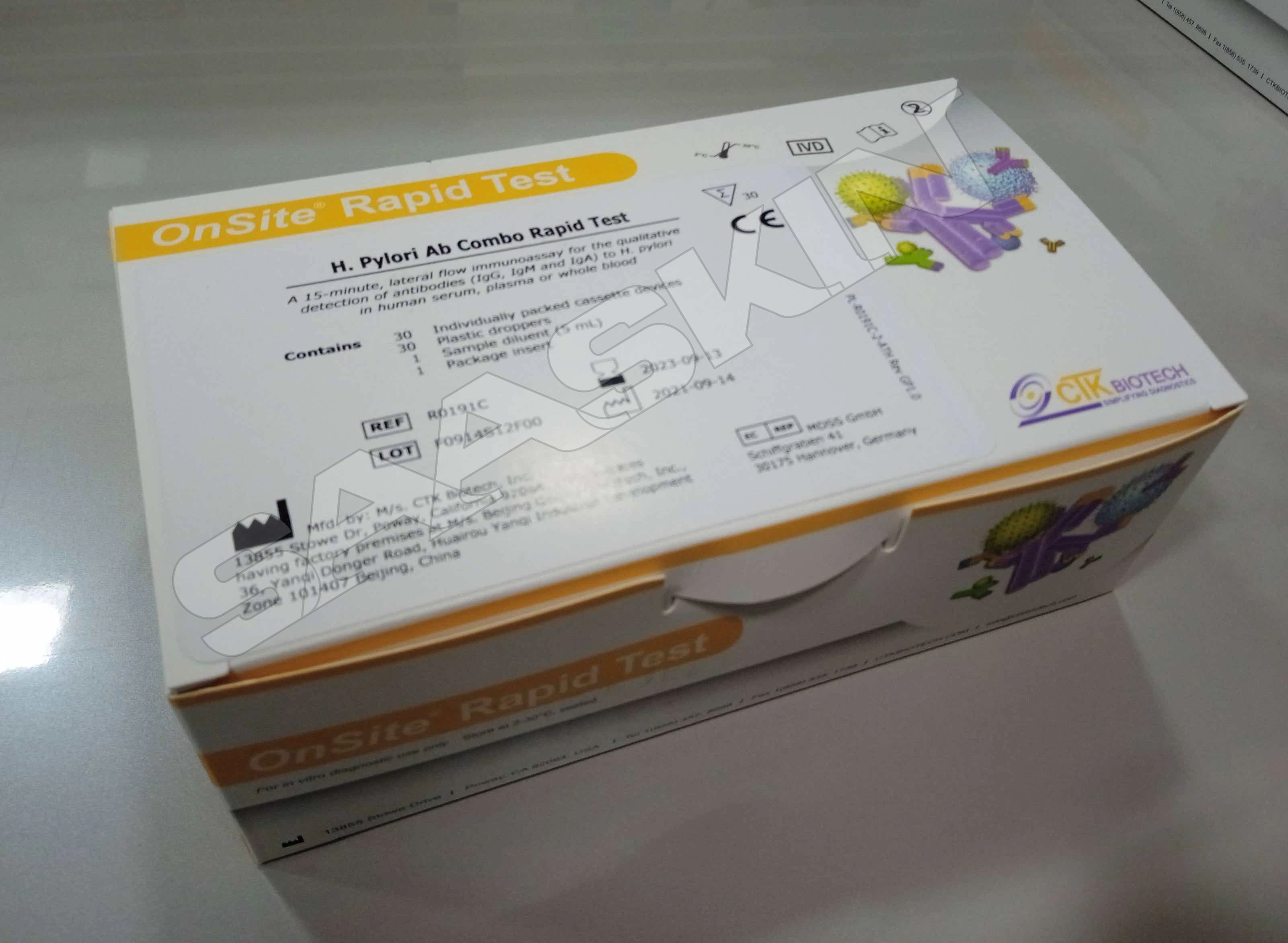
CTK Biotech OnSite H. pylori Ab Combo Rapid Test
Product Details:
- Usage Type Single use
- Features Quick, easy, reliable, qualitative detection
- Measurement Range Qualitative detection, not quantitative
- Accuracy >98%
- Storage Instructions Store at 2-30C, away from sunlight and moisture
- Function Detects IgG, IgA, and IgM antibodies to H. pylori
- Shelf Life 24 months from date of manufacture
- Click to View more
X
CTK Biotech OnSite H. pylori Ab Combo Rapid Test Price And Quantity
- Not included; available separately
- Whole blood, serum, or plasma
- Individually packed in foil pouches, 25 tests per box
- Specified on packaging
- Gastroenterology, Infection screening
- Biohazard waste as per local regulations
- Test cassettes, buffer, instructions for use, droppers
- 10 minutes
- 97.5%
- 98.3%
- Two lines: positive; One line: negative
- Indicated on each test cassette
CTK Biotech OnSite H. pylori Ab Combo Rapid Test Product Specifications
- >98%
- No
- Zero
- Yes
- Store at 2-30C, away from sunlight and moisture
- Silent
- Detects IgG, IgA, and IgM antibodies to H. pylori
- Rapid Test Kit
- Yes
- Individual pouch approx. 9 cm x 5 cm x 0.8 cm
- Plastic test cassette, buffer, dropper, desiccant
- Quick, easy, reliable, qualitative detection
- Visual color line indication
- Qualitative detection, not quantitative
- New
- Detection of H. pylori antibodies
- IVD (In Vitro Diagnostic)
- Manual
- Immunochromatographic assay
- Single use
- White cassette with color indicator lines
- Approx. 15 grams per pouch
- 24 months from date of manufacture
- Not included; available separately
- Whole blood, serum, or plasma
- Individually packed in foil pouches, 25 tests per box
- Specified on packaging
- Gastroenterology, Infection screening
- Biohazard waste as per local regulations
- Test cassettes, buffer, instructions for use, droppers
- 10 minutes
- 97.5%
- 98.3%
- Two lines: positive; One line: negative
- Indicated on each test cassette
CTK Biotech OnSite H. pylori Ab Combo Rapid Test Trade Information
- 100 Unit Per Day
- 1-7 Days
Product Description
| Brand | CTK Biotech |
| Sample Type | Serum/ Plasma/ WB |
| Pack Size | 30T |
| CAT No | R0191C |
The OnSite H. pylori Ab Combo Rapid Test is a sandwich lateral flow chromatographic immunoassay for the qualitative detection of antibodies (IgG, IgM and IgA) against Helicobacter pylori (H. pylori) in human serum, plasma or whole blood.
Rapid and Reliable H. pylori Detection
Engineered for efficiency, the OnSite H. pylori Ab Combo Rapid Test delivers quick, accurate results where they're needed most. Its immunochromatographic technology ensures robust detection of H. pylori antibodies within only 10 minutes, supporting timely clinical decisions and infection management in both clinical and point-of-care settings.
User-Friendly Test Process
Each test kit contains clear instructions, droppers, buffer, and all components required for immediate use. The straightforward visual color indication means only minimal training is needed, making this kit suitable for hospitals, clinics, and laboratories. Its compact, individually-packed cassettes ensure easy handling and safe storage.
Convenient and Safe for Professionals
The tests are designed for single-use and should be disposed of following local biohazard protocols. The kit's shelf life of 24 months ensures long-term reliability, and all cassettes are marked with lot numbers and expiration dates for traceability. External controls are available upon request for enhanced quality assurance.
FAQ's of CTK Biotech OnSite H. pylori Ab Combo Rapid Test:
Q: How is the CTK Biotech OnSite H. pylori Ab Combo Rapid Test used?
A: The test is performed manually by adding a sample of whole blood, serum, or plasma to the cassette, followed by buffer. Results are visually interpreted by the appearance of colored lines within 10 minutes.Q: What types of samples can be used with this rapid test kit?
A: You can use whole blood, serum, or plasma. This flexibility allows clinicians to utilize the specimen type most easily obtained or most appropriate for the patient.Q: When and where should this rapid test be used?
A: The kit is intended for use in clinical settings such as hospitals, clinics, and diagnostic laboratories, and is suitable for point-of-care testing by trained professionals. Testing should be conducted when H. pylori infection is suspected.Q: What is the process for interpreting the test results?
A: Two colored lines indicate a positive result, while a single line in the control region indicates a negative. No lines or only a test line indicates an invalid result and the test should be repeated.Q: What are the key benefits of using this test kit?
A: It delivers rapid, accurate results with high sensitivity and specificity, requires no instrumentation, is portable, and is easy to use, facilitating timely clinical intervention.Q: How should unused tests and used materials be disposed of?
A: Dispose of any used test cassettes and accessories as biohazard waste in accordance with local regulations to ensure safety and compliance with health protocols.Q: Where can users find the expiration date and lot number for quality control?
A: Both the expiration date and lot number are clearly indicated on each individual test cassette and on the outer packaging, ensuring traceability and quality assurance.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
Other Products in 'Medical RTD' category
 |
SAASKIN CORPORATION PRIVATE LIMITED
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |